Frequently Asked Questions
What are the benefits of membrane-based nitrogen removal systems?
Membrane-based systems offer several advantages:
Reduce nitrogen to pipeline specifications
High hydrocarbon recovery, preserving valuable gas components
Low installation cost and quick setup
Minimal maintenance with no moving parts
Flexible and portable systems for different locations
How efficient is nitrogen removal with membrane systems?
Membrane systems are highly efficient, capable of:
Treating gas streams with 4% to 50% nitrogen content
Achieving pipeline-quality gas output
Delivering over 90% hydrocarbon (BTU) recovery
This makes them a reliable and economical solution for improving natural gas quality.
Can CO₂ removal systems handle fluctuations in Natural gas composition?
Yes, membrane-based CO₂ removal systems are highly flexible and can handle variations in natural gas composition and flow rates. Their robust membrane chemistry, flexible configuration and high efficiency membranes allow the system to adapt to changing CO₂ levels without compromising performance, making them suitable for both onshore and offshore applications.
How efficient is membrane-based CO₂ removal from Natural Gas?
Membrane-based CO₂ removal systems are highly efficient, typically achieving over 98+% hydrocarbon recovery, with performance reaching up to more than 98% in optimized systems. They can also reduce CO₂ content to pipeline specifications (often below 1-2%), ensuring high-quality output.
What are the advantages of membrane technology for CO₂ removal from Natural Gas?
Membrane technology offers several advantages, including a compact and cost-effective design, minimal pretreatment requirements, condensation-mode operation and chemical-free operation. These systems are easy to install, require low maintenance, and provide high operational uptime. Additionally, their modular structure allows scalability and flexibility for different project sizes.
Can membranes be applied to EOR applications with high CO2 content?
Yes, membrane-based CO2 removal systems are robust and highly flexible to handle CO2 content in the feed gas as high as 98-99%.
Can CO₂ removal systems handle fluctuations in biogas composition?
Yes, membrane-based CO₂ removal systems are highly flexible and can handle variations in biogas composition and flow rates. Their modular design and high flux membranes allow the system to adapt to changing CO₂ levels without compromising performance, making them suitable for both landfill and biodigester applications.
How efficient is membrane-based CO₂ removal?
Membrane-based CO₂ removal systems are highly efficient, typically achieving over 95% hydrocarbon recovery, with performance reaching up to 98% in optimized systems. They can also reduce CO₂ content to pipeline specifications (often below 2%), ensuring high-quality biomethane output.
What are the advantages of membrane technology for CO₂ removal from biogas?
Membrane technology offers several advantages, including a compact and cost-effective design, minimal pretreatment requirements, and chemical-free operation. These systems are easy to install, require low maintenance, and provide high operational uptime. Additionally, their modular structure allows scalability and flexibility for different project sizes.
Can CO₂ removal systems handle fluctuations in biogas composition?
Yes, membrane systems are specifically designed to manage fluctuations in biogas composition. Their robust membrane chemistry and flexible configuration ensure consistent performance even when CO₂ concentrations or gas flow rates vary, maintaining stable output quality.
How does CO₂ removal from biogas support sustainability goals?
CO₂ removal upgrades biogas into renewable natural gas (RNG) or biomethane, which can be injected into pipelines or used as a clean fuel. This process reduces greenhouse gas emissions, converts waste into valuable energy, and supports circular economy practices by utilizing organic waste streams efficiently.
Why is it essential to remove H₂S from natural gas?
Removing hydrogen sulfide (H₂S) is essential because it is toxic, corrosive, and harmful to equipment and infrastructure. Its presence in natural gas can damage pipelines, processing units, and engines, while also posing serious health and safety risks. Additionally, untreated H₂S can make gas unsuitable for transport or use.
How does H₂S removal improve natural gas quality?
H₂S removal significantly improves natural gas quality by producing a cleaner, safer, and pipeline-compliant gas stream. It reduces corrosion potential, lowers acid gas content, and ensures the gas meets required specifications, making it suitable for commercial use and transportation.
What are common methods used for H₂S removal in natural gas processing?
Common methods for H₂S removal include amine treatment systems, chemical scavengers, and membrane-based separation technologies. In many applications, membrane systems are used for bulk H₂S removal and can be combined with conventional processes for final polishing to meet strict pipeline standards.
How do membrane systems work for H₂S removal?
Membrane systems, such as MTR’s SourSep™, work by separating H₂S from natural gas based on selective permeability. The high-pressure gas stream passes through the membrane, producing:
A sweetened gas stream (low H₂S)
A low-pressure stream rich in H₂S
These systems can achieve significant bulk removal (often up to 75–90% reduction) in a simple, single-stage process without using chemicals or moving parts.
How does H₂S removal impact environmental compliance?
H₂S removal is critical for environmental compliance by reducing harmful emissions and preventing acid gas release into the atmosphere. It helps operators meet regulatory standards for air quality and emissions while supporting safer and more sustainable natural gas processing operations.
How does LPG recovery technology work?
LPG recovery technology combines compression, cooling, membrane separation, and fractionation. The gas stream is first compressed and cooled to condense heavier hydrocarbons (C₃+). It then passes through membranes that selectively separate LPG components from lighter gases. Finally, fractionation produces specification-grade LPG and condensate for use or sale.
Where is LPG extraction technology used?
LPG extraction technology is widely used in applications such as wellhead gas processing, flare gas recovery, dew point control, and refinery or petrochemical streams. It is especially valuable in remote locations where associated gas would otherwise be flared, allowing operators to recover and utilize hydrocarbons locally.
What are the key benefits of modern LPG extraction technology?
Modern LPG extraction technologies offer several benefits, including:
High LPG recovery (up to 95%)
Reduced flaring and lower emissions
Compact, skid-mounted systems for easy deployment
No need for chemicals, absorbents, or cryogenic processes
Quick payback (often within 6–18 months)
These advantages make membrane-based systems efficient, cost-effective, and easy to operate even in remote environments.
Is LPG extraction technology the same as LPG recovery?
Yes, LPG extraction and LPG recovery generally refer to the same process, separating and capturing liquefied petroleum gas (propane, butane, and heavier hydrocarbons) from gas streams. The terms are often used interchangeably in natural gas and refinery applications.
Do LPG extraction technologies require high temperatures or pressures?
No, modern membrane-based LPG extraction systems typically operate at moderate pressures and near-ambient temperatures. They avoid the need for energy-intensive cryogenic processes, making them more efficient and easier to operate in a wide range of conditions.
Where are PerVap membrane separation systems used?
PerVap membrane separation systems are used in food processing and water treatment. Examples of applications include the following:
- Recovery of flavor compounds from food industry process streams.
- Recovery of ethanol from fermentation and food industry process streams.
- Removal of organic contaminants from wastewater streams.
How do PerVap® systems work?

Pervaporation process
In the pervaporation process, a liquid mixture contacts the membrane, which preferentially permeates one of the liquid components as a vapor. The vapor enriched in the more permeable component is cooled and condensed, spontaneously generating a vacuum that drives the process. MTR’s pervaporation systems use proprietary membranes to separate dissolved volatile organic compounds (VOCs) from aqueous solutions. The membrane selectively permeates the organic compound, producing a concentrated permeate stream that contains the organic component of interest and an organic-depleted residue stream. The dissolved organic may be a high-value component, such as a food essence, or a contaminant, such as a chlorinated solvent.
What is the payback time for a VaporSep membrane system?
The payback time is typically 6 to 12 months, depending on the application.
What is the lifetime of a VaporSep membrane?
With proper operation and maintenance, a VaporSep membrane should have a lifetime of 3 to 5 years. When replacement is necessary, the membrane module is easily replaced without the need for special tools or expertise.
What is the footprint of a VaporSep membrane system? How much does it weigh?
Large membrane systems may consist of multiple skids and subsystems, such as a compressor package, a dryer package, and a refrigeration package. The footprint of a skid may be as large as 30 feet long by 12 feet wide. Since every system is custom designed, the footprint may be reduced to fit the customer’s requirements. Individual skids weigh as much as 25 tons.
What is a typical turndown ratio?
Membrane systems can be operated down to 20% of design capacity.
What process controls and instrumentation are required?
MTR will provide a standard piping and instrumentation diagram (P&ID) showing recommended control systems and instruments for the membrane and pretreatment equipment. MTR can review and approve customized control systems for special applications.
What preconditioning of the feed gas is required?
Several factors determine the required pretreatment. If dust or other solid particles are present, they must be removed upstream with a high-efficiency filter. If the feed gas contains entrained liquids, a mist eliminator vessel with a high-efficiency coalescing agent is required. MTR will provide detailed pretreatment requirements.
What are the operating limits of VaporSep membrane modules?
The membrane module can operate over a wide range of temperatures and pressures. The temperature range is -40°C to 40°C. The feed pressure can be as high as 1,500 psi. The permeate pressure can be under vacuum if required to achieve a certain separation.
Where are VaporSep membrane separation systems used?
VaporSep membrane separation systems are used in the petrochemical, refining, and natural gas processing industries. Current applications include the following:
- Recovery of olefins from resin degassing vent streams in polyolefin plants.
- Recovery of liquified petroleum gas (LPG) from refinery vent streams.
- Fuel gas conditioning (removal of heavy hydrocarbons from fuel gas).
- Recovery of natural gas liquids (NGLs) from natural gas streams.
How are VaporSep membranes packaged?
VaporSep membranes are manufactured as flat sheets and rolled into spiral-wound modules. The feed gas enters the module and flows between the membrane sheets. Spacers on the feed side and the permeate side of the membrane sheets create flow channels. The hydrocarbon vapor that passes preferentially through the membrane flows inward to a central permeate collection pipe. The light gas (nitrogen, hydrogen or methane) is rejected by the membrane and exits as the residue.
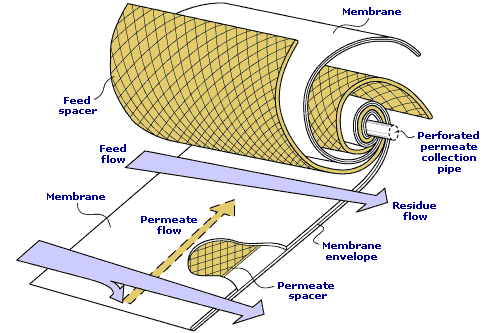
Spiral-wound membrane module
Modules are 3 feet long and 4 to 8 inches in diameter. As many as 4 modules are placed in pressure vessels designed to meet local standards (ASME, etc). To meet the needs of a particular application, modules are configured in series and parallel flow combinations.
How do VaporSep membranes work?
VaporSep membranes separate gas mixtures on the basis of solubility. Large hydrocarbon molecules with greater solubility in the membrane permeate much faster than smaller, less soluble molecules such as nitrogen, hydrogen, or methane.
By comparison, conventional membranes separate gases on the basis of size. Small molecules are selectively permeated because they diffuse through the membrane more rapidly than large molecules.
How does the VaporSep® process work?
The VaporSep process combines a compression-condensation step with a membrane separation step. The feed gas – a mixture of hydrocarbons in nitrogen, hydrogen, or methane – is compressed and cooled, condensing a portion of the hydrocarbons in the gas. The liquid hydrocarbons are recovered; the remaining gas, which still contains significant amounts of hydrocarbons, is fed to the VaporSep membrane. The membrane separates the gas into a hydrocarbon-rich permeate stream and a hydrocarbon-depleted residue stream (the purified gas). The permeate is recycled to the compressor; the residue stream is vented or reused.
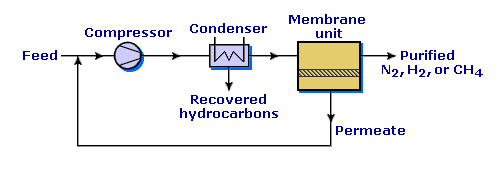
VaporSep process
When my company invests in a membrane system from MTR, what does it get?
For each application, MTR:
- Manufactures membranes and spiral-wound modules.
- Selects and procures all system components, including compressors, pumps, heat exchangers, vessels, instruments, and controls.
- Designs piping and system layout.
- Supervises skid fabrication.
- Inspects and tests the equipment before shipment.
- Trains the operators and commissions the system.
- Guarantees the performance of the entire system.
In summary, MTR does everything from initial concept to commissioning except the field installation work.
How do I find out if a membrane separation can improve the economics of my process?
Contact us by phone, by e-mail, or submit your request using one of our online inquiry forms. We will get back to you as soon as possible.
- Telephone your local sales agent.
- Scan a diagram and send it as an e-mail attachment together with a description of your process to sales@mtrinc.com.
- Select and complete one of our online inquiry forms on the Contact Us page.
What are the benefits of including an MTR membrane system in my process?
MTR’s membrane systems enable operators to do the following:
- Recover valuable raw materials, such as olefins.
- Purify and recycle purge gases, such as nitrogen and hydrogen.
- Reduce emissions.
What is membrane separation and how does it work?
Membrane separation is a physical process that uses a semi-permeable membrane to selectively separate components in a gas or liquid mixture based on differences in molecular size, charge, or chemical affinity. A pressure difference across the membrane acts as the driving force, allowing certain molecules to pass through (permeate) while retaining others (retentate). This technology is widely used in industries like petrochemical, natural gas processing, and water treatment for applications ranging from gas purification to solvent recovery.
What types of membrane separation techniques are there? (e.g., UF, NF, RO)
The main types of membrane separation techniques include microfiltration (MF) for removing suspended solids and bacteria, ultrafiltration (UF) for filtering proteins and macromolecules, nanofiltration (NF) for removing dissolved organics and divalent ions, and reverse osmosis (RO) for desalination and removing virtually all dissolved particles. Additionally, gas separation membranes are used in industrial applications such as hydrogen recovery, CO₂ removal, and hydrocarbon recovery from petrochemical and natural gas process streams.
How does pressure affect membrane separation performance?
Pressure is the primary driving force in membrane separation, as it pushes molecules through the membrane’s selective barrier. Higher applied pressure generally increases the permeation rate (flux), allowing more material to pass through the membrane per unit time. However, excessively high pressure can accelerate membrane fouling and reduce membrane lifespan, so operating pressure must be carefully optimized for each application.
How to choose the right membrane for a specific separation task?
Choosing the right membrane depends on several key factors: the size and type of molecules you need to separate, the required purity level of the product, and the operating conditions such as temperature, pressure, and chemical compatibility. You should also consider the feed composition, desired recovery rate, and whether you need to handle aggressive chemicals like H₂S or CO₂. Consulting with a membrane technology specialist, such as MTR (Membrane Technology and Research), can help match the optimal membrane material and system design to your specific process requirements.
Difference between membrane separation and traditional separation methods?
Membrane separation is a purely physical process with no phase change, no chemical additives, and no heat required, making it simpler and more energy-efficient than traditional methods like distillation, absorption, or cryogenic separation. Traditional methods often involve large equipment footprints, high energy consumption, and complex operations with moving parts. Membrane systems are compact, modular, easy to install, and can operate unattended, making them ideal for remote or offshore locations.
What are the applications of membrane separation?
Membrane separation is used across a wide range of industries including petrochemical plants for hydrocarbon and monomer recovery, natural gas processing for CO₂ and H₂S removal, refineries for hydrogen purification, and water treatment for desalination and wastewater reuse. Companies like MTR (Membrane Technology and Research, Inc.) have deployed over 450 membrane gas separation systems worldwide for applications including polyethylene production vent recovery, fuel gas conditioning, nitrogen removal, and solvent dehydration.
What is a hydrocarbon recovery system and how does it work?
A hydrocarbon recovery system captures valuable hydrocarbons, such as ethylene, propylene, and other monomers, from industrial vent and purge gas streams that would otherwise be flared or lost to the atmosphere. These systems typically use membrane technology (such as MTR’s VaporSep®) to separate hydrocarbons from light gases like nitrogen and hydrogen based on differences in permeation rates through the membrane. The recovered hydrocarbons are recycled back into the production process, reducing raw material costs and improving plant profitability.
Types of hydrocarbon recovery systems used in the oil & gas industry.
The main types of hydrocarbon recovery systems used in the oil and gas industry include membrane-based separation systems, cryogenic recovery units, absorption-based systems (using lean oil or refrigerated solvents), and pressure swing adsorption (PSA). Membrane systems like MTR’s VaporSep® are increasingly preferred for petrochemical applications because they are compact, have no moving parts, require minimal maintenance, and offer fast payback, typically less than one year.
What problems does a hydrocarbon recovery system solve?
Hydrocarbon recovery systems solve the problem of valuable feedstock being wasted through flaring or venting at petrochemical plants, refineries, and gas processing facilities. In polyethylene production alone, feedstock losses from distillation column overheads, reactor purges, and resin degassing vents can range from $1 million to $3 million per year per plant. These systems recover that lost value while also addressing environmental compliance requirements by reducing volatile organic compound (VOC) emissions and flare loads.
How hydrocarbon recovery helps reduce flaring and emissions?
Hydrocarbon recovery systems directly reduce flaring by capturing hydrocarbons from vent streams and recycling them back into the process instead of burning them at the flare. This significantly lowers CO₂, methane, and other greenhouse gas emissions associated with routine flaring. For example, MTR’s membrane-based VaporSep® systems recover more than 90% of vent hydrocarbons, which drastically cuts the volume of gas sent to the flare and helps operators meet increasingly strict environmental regulations.
What are the main components of a hydrocarbon recovery system?
The main components of a membrane-based hydrocarbon recovery system include a compressor to pressurize the feed gas, a condenser and gas/liquid separator to remove condensable hydrocarbons, membrane modules that selectively separate hydrocarbons from light gases, and associated piping, instrumentation, and controls. MTR’s complete VaporSep® units are skid-mounted for easy installation, handling vent streams from 300 to 30,000 lb/h with hydrocarbon concentrations of 10 to 80 vol%.
What is a propylene recovery unit and how does it work?
A propylene recovery unit (PRU) is a system used in polypropylene production plants to recover valuable propylene that is otherwise lost in vent streams.
It works by first compressing and cooling the vent gas to condense part of the propylene. The remaining gas is then passed through a membrane system, which separates it into two streams:
A propylene-rich stream, which is recycled back into the process
A nitrogen-rich stream, which is purified and reused
This process enables efficient recovery and reuse of both propylene and nitrogen within the plant.
How does a propylene recovery unit improve process efficiency?
A propylene recovery unit improves efficiency by minimizing the loss of valuable raw materials and reducing waste.
By recovering up to 99% of propylene, the system significantly lowers feedstock costs and increases overall plant profitability. It also reduces flaring and emissions while allowing nitrogen to be reused in the process. Additionally, membrane systems can achieve higher recovery rates than traditional condensation alone, improving overall process performance.
What technologies are used inside a propylene recovery unit?
A propylene recovery unit typically combines multiple technologies, including:
Compression systems – to increase pressure for effective separation
Condensation units – to recover bulk propylene as liquid
Membrane separation technology (e.g., VaporSep®) – to separate remaining propylene from nitrogen
Heat exchangers and control systems – for temperature control and process optimization
The membrane system is the core technology, enabling selective separation of propylene from lighter gases without the need for chemicals or complex operations.
How to optimize the efficiency of a propylene recovery unit in a refinery?
To optimize efficiency, operators should focus on:
Maintaining proper pressure and temperature conditions for optimal separation
Ensuring membrane performance and cleanliness through regular monitoring
Optimizing recycle streams to maximize propylene recovery
Integrating membrane systems with existing compression and condensation stages
Monitoring feed composition and adjusting operating parameters accordingly
Combining condensation with membrane separation is especially effective, as it enables higher recovery rates even at moderate conditions.
How to select the right technology for a propylene recovery unit?
Selecting the right technology depends on several factors:
Feed composition and flow rate
Required recovery efficiency (e.g., 90% vs. 99%+)
Operating conditions (pressure, temperature)
Space and installation constraints
Cost and payback period
Membrane-based systems are often preferred because they are compact, energy-efficient, and capable of achieving high recovery with fast payback. They also offer easier installation and lower maintenance compared to more complex alternatives.
What is EO and VAM production and how are they related?
Ethylene Oxide (EO) and Vinyl Acetate Monomer (VAM) are important petrochemical products made using ethylene as a key raw material.
EO is produced by reacting ethylene with oxygen, while VAM is produced by reacting ethylene with oxygen and acetic acid. Both processes operate in similar reactor loop systems and share a common challenge — loss of valuable ethylene in purge gases.
How does EO and VAM production work in petrochemical plants?
In both EO and VAM production:
Ethylene reacts with oxygen (and acetic acid in VAM) in a catalytic reactor
Only part of the ethylene converts in one pass, so unreacted gases are recycled
Inert gases like argon build up in the loop and must be purged
This purge stream contains valuable ethylene, which is often recovered using membrane systems and recycled back into the process
This loop-based process improves efficiency but requires recovery systems to minimize losses.
What factors affect efficiency in EO and VAM production?
Several factors influence efficiency, including:
– Catalyst performance and selectivity (affects conversion rates)
– Reaction temperature and pressure control
– Feedstock purity (ethylene and oxygen quality)
– Recycling and purge gas management
– Ethylene recovery systems (like membranes)
Poor control or losses in purge streams can significantly increase operating costs due to lost ethylene.
What are the main uses of EO and VAM?
Ethylene Oxide (EO):
Production of ethylene glycol (used in polyester and antifreeze)
Surfactants, solvents, and sterilization agents
Vinyl Acetate Monomer (VAM):
Adhesives and coatings
Paints and sealants
Packaging and textile materials
Both are essential building blocks in the chemical and manufacturing industries.
How to control temperature in EO and VAM production units?
Temperature control is critical because the reactions are highly exothermic (heat-generating).
It is managed by:
Using multitubular reactors with cooling systems
Circulating coolants or generating steam to remove excess heat
Maintaining controlled operating ranges (typically ~200–300°C for EO)
Monitoring continuously to avoid side reactions and safety risks
Proper temperature control ensures high selectivity, safety, and consistent product quality.
What are gas recovery systems and how do they work?
Gas recovery systems are technologies used to capture and reuse valuable gases that would otherwise be wasted or flared in industrial processes. In petrochemical plants, off-gas streams often contain a mixture of hydrocarbons and nitrogen.
Modern systems, such as membrane-based solutions, work by separating gases based on their permeability. The gas is compressed and cooled, then passed through membranes, where hydrocarbons permeate faster than nitrogen, producing a hydrocarbon-rich stream that can be reused as fuel while nitrogen is removed.
Why are gas recovery systems important in industrial plants?
Gas recovery systems are critical because they help industries reduce waste, improve efficiency, and lower operating costs. Without recovery systems, valuable hydrocarbons are often burned in flare systems, resulting in energy loss and emissions.
By recovering these gases, plants can convert waste streams into usable fuel, reduce dependency on purchased energy, and minimize environmental impact. This also supports better resource utilization and overall process optimization.
How to select the right gas recovery system for your facility?
Selecting the right gas recovery system depends on several factors, including gas composition, flow rate, pressure, and desired product quality. Facilities should evaluate whether their streams contain recoverable hydrocarbons and define their recovery goals.
Membrane-based systems are often preferred because they are compact, easy to install, and require minimal maintenance. Custom-designed solutions tailored to specific process conditions ensure optimal performance and maximum recovery efficiency.
How efficient are modern gas recovery systems?
Modern gas recovery systems, especially membrane-based technologies, are highly efficient and capable of recovering a significant portion of valuable hydrocarbons. These systems can produce fuel gas with low nitrogen content and high hydrocarbon concentration, making it suitable for reuse.
They also enhance overall plant efficiency by reducing hydrocarbon losses and improving fuel utilization, often delivering strong economic returns with simple operation and low energy requirements.
Why is VCM recovery important in PVC production?
VCM recovery is critical because a significant amount of unreacted vinyl chloride monomer (VCM) is lost in vent streams during PVC production. These emissions are strictly regulated and often require incineration before release. Recovering VCM not only ensures environmental compliance but also allows manufacturers to reuse valuable raw material, improving overall process efficiency and reducing operating costs.
How does membrane-based VCM recovery technology work?
Membrane-based systems, such as VaporSep®, separate VCM from vent gases by allowing VCM to pass through the membrane faster than inert gases. This creates a VCM-rich stream (permeate) that is recycled back into the process, while the remaining gas (residue) is treated or incinerated. The recovered VCM is then condensed and reused in production, creating a closed-loop recovery system.
What are the main benefits of VCM recovery systems?
VCM recovery systems offer several key benefits:
Recover up to 90–99%+ of lost VCM
Reduce raw material losses and improve profitability
Minimize emissions and regulatory risks
Lower energy requirements compared to traditional methods
Compact, low-maintenance systems with no moving parts
These advantages make VCM recovery both economically and operationally efficient.
What is the best technology for VCM recovery in PVC plants?
Membrane-based recovery systems are considered one of the most effective technologies for VCM recovery. Compared to conventional condensation alone, membranes achieve higher recovery rates and operate efficiently at moderate temperatures and pressures. They also offer a compact design and lower maintenance requirements, making them ideal for modern PVC plants.
How does VCM recovery support sustainability goals?
VCM recovery supports sustainability by reducing harmful emissions, lowering the need for incineration, and minimizing waste. By recycling unreacted VCM back into the production process, it reduces the demand for fresh raw materials and improves resource efficiency. This leads to a cleaner, more sustainable PVC manufacturing process with a smaller environmental footprint.
Why is natural gas treatment important?
Natural gas treatment is essential to remove impurities such as CO₂, H₂S, nitrogen, and heavy hydrocarbons so the gas meets pipeline and operational specifications. Untreated gas can reduce heating value, cause corrosion, and lead to operational issues. Proper treatment ensures safe transportation, improved efficiency, and compliance with industry standards.
Can natural gas treatment be customized for different gas compositions?
Yes, natural gas treatment systems can be fully customized based on the composition of the raw gas and project requirements. Membrane-based solutions are particularly flexible and can be tailored to handle varying levels of CO₂, H₂S, nitrogen, and hydrocarbons to achieve optimal performance and meet specific pipeline or processing standards.
Can natural gas treatment help with dehydration?
Yes, natural gas treatment can also assist in removing water vapor from the gas stream. Membrane systems and integrated treatment processes can reduce moisture content along with other contaminants, helping prevent corrosion, hydrate formation, and operational inefficiencies in pipelines and equipment.
What technologies are commonly used in natural gas treatment?
Common technologies include membrane separation, amine treatment, adsorption (PSA), and cryogenic processes. Among these, membrane technology is widely used due to its simplicity, low operating cost, compact design, and ability to remove multiple contaminants in a single step.
How does natural gas treatment improve gas value?
Natural gas treatment increases the value of gas by removing non-combustible and harmful components like CO₂ and nitrogen, which lowers BTU value. By upgrading gas quality to pipeline standards and recovering valuable hydrocarbons, treated gas becomes more marketable and efficient for end-use applications.
What role do membranes play in natural gas treatment?
Membranes play a key role by selectively separating impurities such as CO₂, H₂S, nitrogen, and heavy hydrocarbons from natural gas streams. These systems are quick to install, easy to operate, require minimal maintenance, and provide high efficiency with strong hydrocarbon recovery, making them a reliable and cost-effective solution for modern gas processing.
Why is fuel gas conditioning necessary for gas engines and turbines?
Fuel gas conditioning is necessary because raw gas often contains impurities such as H₂S, CO₂, nitrogen, and heavy hydrocarbons (C₃+) that can damage engines and turbines. These contaminants can cause corrosion, carbon buildup, and reduced fuel quality, leading to poor performance or even making the gas unusable. Proper conditioning ensures the gas meets required quality standards for safe and efficient operation.
How does fuel gas conditioning benefit my operations?
Fuel gas conditioning improves operational efficiency by enhancing fuel quality and protecting equipment. It helps reduce maintenance costs, prevent unscheduled downtime, and improve the reliability of gas engines and turbines. Additionally, it eliminates the need for alternative fuels like diesel and ensures consistent performance in demanding environments.
What types of technologies are used in fuel gas conditioning?
Fuel gas conditioning commonly uses membrane-based separation technology, such as FuelSep™ systems. These membranes selectively remove impurities like CO₂, H₂S, nitrogen, water, and heavy hydrocarbons from the gas stream. The systems are compact, have no moving parts, and operate efficiently under ambient conditions, making them easy to install and maintain.
Can fuel gas conditioning recover valuable liquids from raw gas?
Yes, fuel gas conditioning can recover valuable hydrocarbons (such as C₃+ liquids) from the raw gas stream. Instead of being wasted, these components are separated and returned to the main gas stream for downstream recovery, helping maximize resource utilization and improve overall process economics.
Does fuel gas conditioning help reduce emissions?
Yes, fuel gas conditioning significantly reduces emissions. By removing heavy hydrocarbons and acid gases like CO₂ and H₂S, it lowers harmful exhaust emissions and helps maintain compliance with environmental regulations. It also reduces non-methane hydrocarbon (NMHC) emissions and minimizes environmental impact.
Why is nitrogen removed from natural gas?
Nitrogen is removed because it reduces the heating value (BTU) of natural gas, making it less efficient and often unsuitable for pipeline transport or sale. High nitrogen content dilutes the energy content, so removing it ensures the gas meets commercial and pipeline quality standards.
What are the common methods for nitrogen removal?
The most common methods for nitrogen removal from natural gas include:
Cryogenic distillation – effective but complex and expensive
Pressure Swing Adsorption (PSA) – suitable for certain conditions
Membrane separation – a simpler, cost-effective solution, especially for small to mid-scale operations
What nitrogen levels are acceptable for pipelines?
Most natural gas pipelines require nitrogen content to be below approximately 4% to 6% to meet quality specifications. Gas exceeding this limit must be treated before it can be transported or sold.