MTR is the leading supplier of membrane system for hydrocarbon recovery from petrochemical plant vents.
Ethylene Oxide (EO) and Vinyl Acetate Monomer (VAM) Production
Opportunity
In ethylene oxide (EO) production, oxygen and ethylene are reacted to form EO. Since the conversion per pass is not 100%, the reactor is operated in loop mode. Argon, which enters with the oxygen feed, builds up in the reactor loop and must be purged. The vinyl acetate monomer (VAM) process is very similar except acetic acid is also fed to the reactor. A portion of the ethylene feedstock is lost with the argon purge gas from the reactor loop. The value of the lost feedstock is substantial — approximately $500,000 per year for a typical EO or VAM plant.
VaporSep® Solution
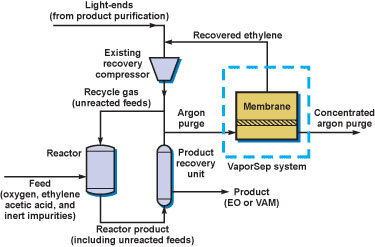
Simplified EO or VAM production process showing addition of the VaporSep® membrane system
The VaporSep unit consists of a single-stage membrane system that separates ethylene vapors from argon. The ethylene-enriched permeate is sent to an existing compressor, then returned to the reactor; the argon-enriched residue is flared.
VaporSep systems recover more than 90% of vent ethylene that would otherwise be flared. VaporSep units are currently used by major EO producers including Samsung, Total, PEMEX and Formosa Plastics and VAM producers including Samsung BP and Dairen.
Frequently Asked Questions
What is EO and VAM production and how are they related?
Ethylene Oxide (EO) and Vinyl Acetate Monomer (VAM) are important petrochemical products made using ethylene as a key raw material.
EO is produced by reacting ethylene with oxygen, while VAM is produced by reacting ethylene with oxygen and acetic acid. Both processes operate in similar reactor loop systems and share a common challenge — loss of valuable ethylene in purge gases.
How does EO and VAM production work in petrochemical plants?
In both EO and VAM production:
Ethylene reacts with oxygen (and acetic acid in VAM) in a catalytic reactor
Only part of the ethylene converts in one pass, so unreacted gases are recycled
Inert gases like argon build up in the loop and must be purged
This purge stream contains valuable ethylene, which is often recovered using membrane systems and recycled back into the process
This loop-based process improves efficiency but requires recovery systems to minimize losses.
What factors affect efficiency in EO and VAM production?
Several factors influence efficiency, including:
– Catalyst performance and selectivity (affects conversion rates)
– Reaction temperature and pressure control
– Feedstock purity (ethylene and oxygen quality)
– Recycling and purge gas management
– Ethylene recovery systems (like membranes)
Poor control or losses in purge streams can significantly increase operating costs due to lost ethylene.
What are the main uses of EO and VAM?
Ethylene Oxide (EO):
Production of ethylene glycol (used in polyester and antifreeze)
Surfactants, solvents, and sterilization agents
Vinyl Acetate Monomer (VAM):
Adhesives and coatings
Paints and sealants
Packaging and textile materials
Both are essential building blocks in the chemical and manufacturing industries.
How to control temperature in EO and VAM production units?
Temperature control is critical because the reactions are highly exothermic (heat-generating).
It is managed by:
Using multitubular reactors with cooling systems
Circulating coolants or generating steam to remove excess heat
Maintaining controlled operating ranges (typically ~200–300°C for EO)
Monitoring continuously to avoid side reactions and safety risks
Proper temperature control ensures high selectivity, safety, and consistent product quality.